Acid-catalyzed Hydration of 1-butene Will Produce Which of the Following
In the first step there happens proton transfer in which hydrogen gets transfered to terminal carbon that result in the formation of carbocation. Show transcribed image text.
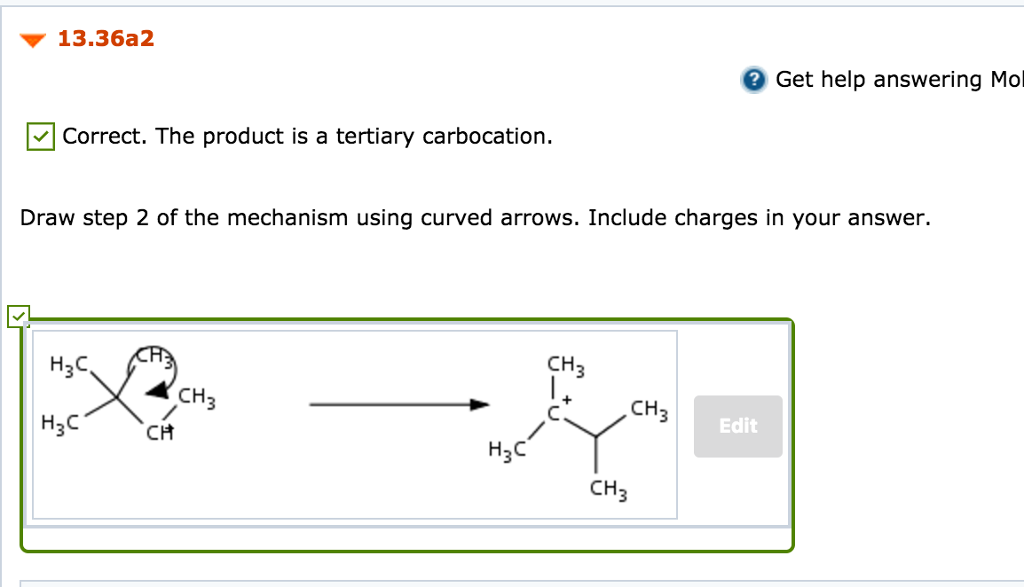
Solved Practice Problem 13 36 Acid Catalyzed Hydration Of Chegg Com
View solution The product obtained in the above reaction is.
. Science Chemistry QA Library Acid-catalyzed hydration of 2-Methyl-1-butene yields two alcohols. For each of the four steps add one or two curved arrows to the. 3-Methyl-1-butene C5H10 CID 11239 - structure chemical names physical and chemical properties classification patents literature biological activities.
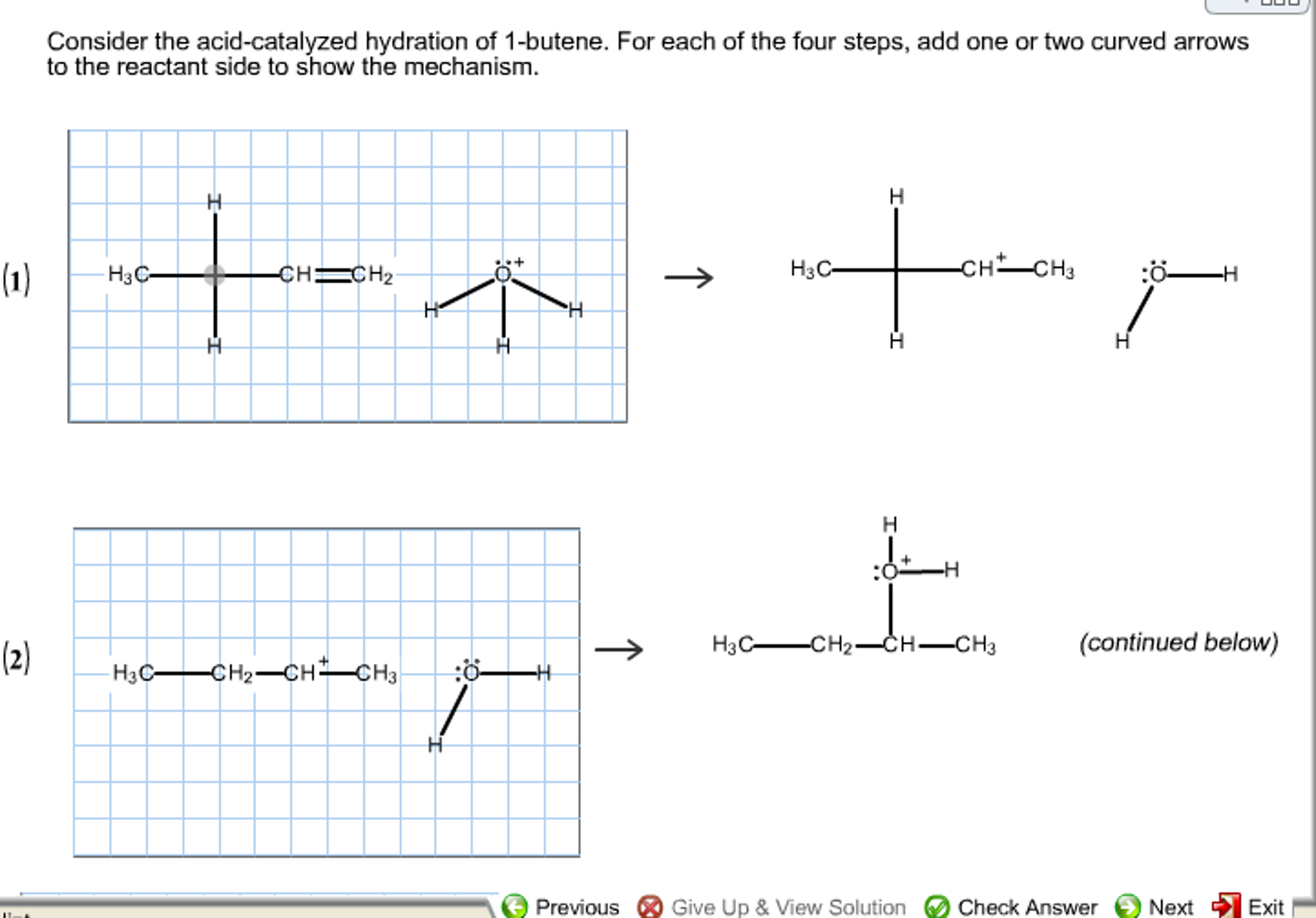
Consider the acid-catalyzed hydration of 3-methyl-1-butene. Acid-catalyzed hydration of 2-methyl-1-butene would yield which of the following. Which type of alcohol is formed by the acid catalyzed hydration of 1 butene.
Acid-catalyzed hydration of 33-dimethyl-1-butene produces 23-dimethyl-2-butanol. 1-hexene Z-2-hexene E-2-hexene 2-methyl-1-pentene 23-dimethyl-2. Science Chemistry QA Library The acid-catalyzed hydration of 33-dimethyl-1-butene gives 23-dimethyl- 2- butanol as the major product.
For the following reaction. This is done by adding an alcohol to the more substituted carbon atom and hydrogen to the less substituted carbon atom. Acid catalyzed hydration of alkenes involves replacing the pi bond on an alkene with a water molecule.
By adding an alcohol to one of the more than substituted carbon atoms and hydrogen to another carbon atom there is a larger-than-lipophilic substitution for one of the subatomic carbons. 1 Add curved arrows for the first step. View solution Write IUPAC names of the products.
2 Draw both the organic and inorganic intermediate species. Acid-catalyzed hydration of 2-methyl-1-butene would yield which of the following. The major product does not undergo oxidation while the minor product will undergo oxidation.
Mention stereo chemistry where applicable Medium. The acid catalyzed hydration of 2-methyl-2-butene. DRAW STEP 3 see picture.
The major product is 2-methylbutan-2-ol. The selective hydrogenation of 2-butyne-14 diol to cis-2-butene-14 diol has been studied using different palladium-supported catalysts. This problem has been solved.
What will be the product formed by hydrogenation of 2 Butyne. OH HO OH 16. False the acid-catalyzed hydration of both 1- and 2-butene gives 2-butanol.
Markovnikovs rule would be followed by. Arrange the following alkenes according to their reactivity toward acid-catalyzed hydration. Acid-catalyzed hydration of 2-methyl-1-butene would yield which of the following.
Which of the following alcohols is produced by the acid catalyzed hydration of 2-methyl- 2-butene. Which of the following on acid catalysed hydration produce mainly. Propose a mechanism for the formation of this alcohol.
October 30 2018 By Leah4sci 1 Comment. Include nonbonding electrons and charges where applicable. In the second step there happen methyl shift to convert secondary carbocation to tertiary carboocation which is more stable.
In the 3rd step there happens nucleophilic attack by. The yields for an acid catalyzed hydration of an alkene can be increased by using dilute acid as this will help drive the equilibrium to products. The acid-catalyzed hydration of 33-dimethyl-1-butene gives 23-dimethyl- 2-butanol as the major product.
Which one of the following alcohols would dehydrate most rapidly when treated with. This reaction follows Markovnikovs rule and may undergo a carbocation rearrangement. Chemistry questions and answers.
See the answer See the answer done loading. Propose a mechanism for the formation of this alcohol. True Due to the formation of a mercurinium ion in hydration of an alkene using oxymercuration- demercuration we see overall anti-product form making this reaction stereospecific.
True or false alkynes are always more acidic than alkenes. Which alcohol would initially produce the most stable carbocation when treated with. Which of the following alcohols is produced by the acid catalyzed hydration of 2-methyl- 2-butene.
Outline a detailed mechanism for the acid catalyzed hydration of 2-methyl-2-butene and name the major product according to the IUPAC rules. Include arrows charges and lone pairs. Which alkene would you expect to be most reactive towards acid-catalysed hydration.
Acid-catalyzed dehydration of 33-dimethyl-2-butanol is the rate determining step. Acid-catalyzed hydration of 2-methyl-1-butene would yield which of the following. Taking an alkenes pi bond replacing it with a water molecule is how acid catalyzed hydration is performed.
View solution Identify the products in the following reaction. Explain why by showing the structures of the products. False although the C- conjugate base of an alkyne is sp-hybridized and thus more stable than that of an alkene which sp2- hybridized this is true only if there are no other effects stabilizing the conjugate base of alkene.
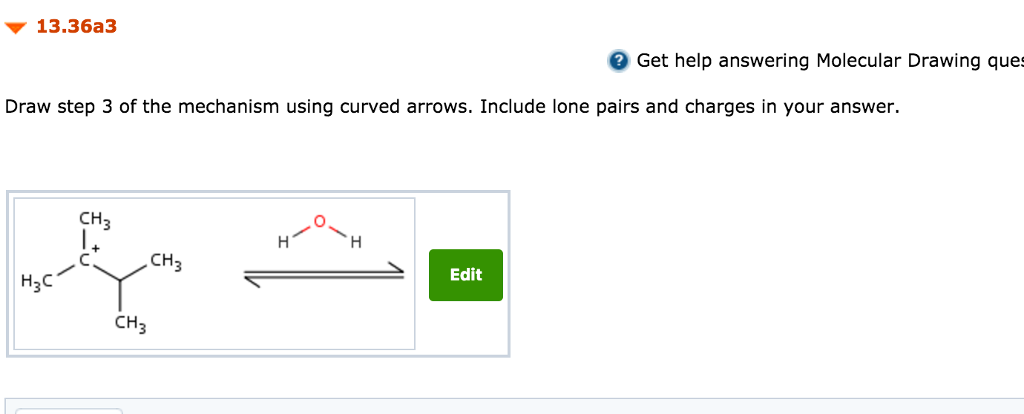
Solved Practice Problem 13 36 Acid Catalyzed Hydration Of Chegg Com

Solved Consider The Acid Catalyzed Hydration Of 1 Butene Chegg Com
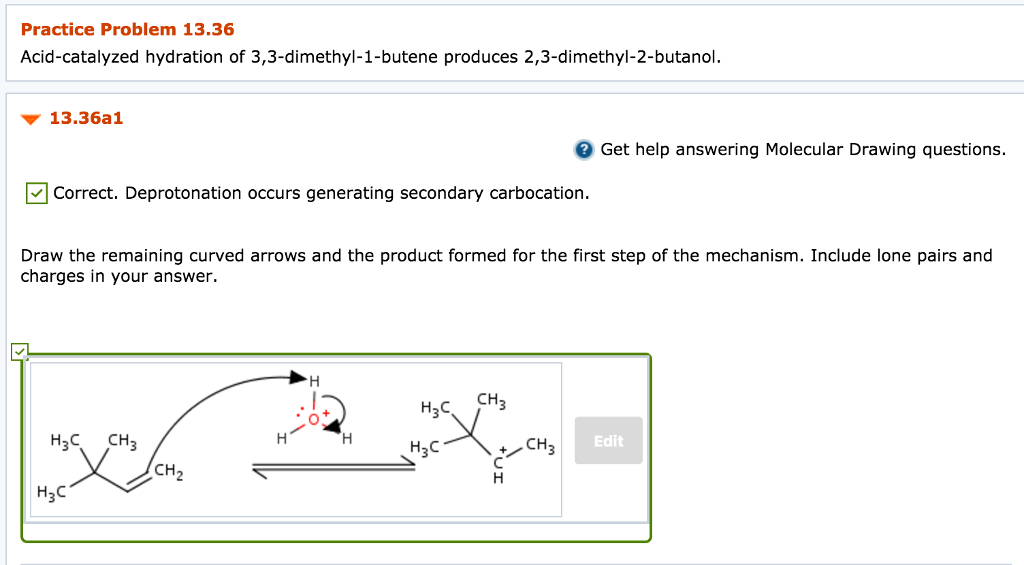
Solved Practice Problem 13 36 Acid Catalyzed Hydration Of Chegg Com
Comments
Post a Comment